Examining the efficacy of lispro in postprandial glucose control for diabetes
Despite the range of available antihyperglycemic therapies, 57%–68% of Chinese patients with T2D fail to attain glycated hemoglobin A1c (HbA1c) target levels of <7.0%. Since PPG levels have better predictive value for cardiovascular disease risk and all-cause mortality compared with FPG levels, and 2-h PPG levels have a stronger correlation with cardiovascular events than HbA1c levels, the importance of targeting PPG has been recognized in recent guidance on insulin therapy.
However, the action of many bolus insulins is not sufficiently rapid to match carbohydrate absorption, limiting their efficacy and dosing flexibility. Ultra rapid lispro (URLi) is a novel formulation of insulin lispro designed to more closely match the physiological insulin response to a meal, with the aim of improving postprandial glucose (PPG) control.
This study was led by Prof. Weiping Jia (Department of Endocrinology and Metabolism, Shanghai Jiao Tong University School of Medicine Affiliated Sixth People’s Hospital).This study was a multinational, multicenter, randomized, double-blind, treat-to-target, 26-week, phase 3 trial to evaluate the efficacy and safety of URLi in adults with type 2 diabetes (T2D). Patients were recruited across 41 sites in China, Argentina, and Mexico.
A total of 595 eligible participants were randomized and 481 patients (80.8%) were Asian (Chinese). After an 8-week lead-in period during which basal insulin glargine or degludec was optimized, adults with T2D were randomized (2:1) to prandial URLi (n = 395) or lispro (n = 200). The primary endpoint was non-inferiority of URLi versus lispro in glycated hemoglobin A1c (HbA1c) change from baseline to week 26. Multiplicity-adjusted analyses were performed to assess the superiority of URLi in 1- and 2-h PPG excursions during a mixed-meal tolerance test (MMTT) and HbA1c change at week 26.
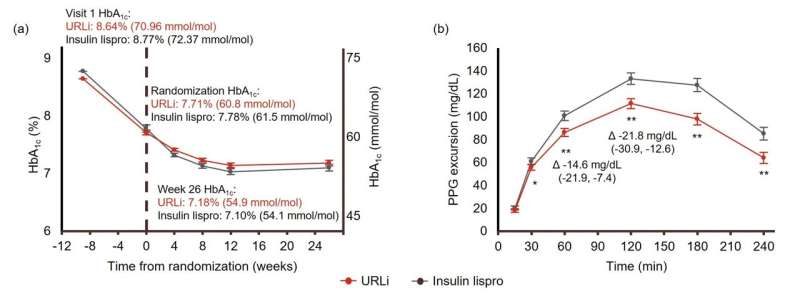
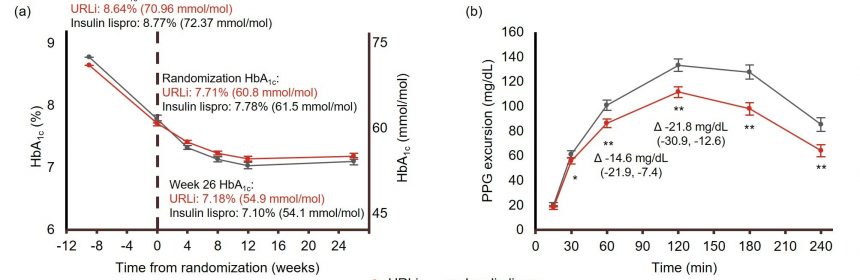
URLi showed non-inferiority for HbA1c change at week 26 versus lispro (least-squares mean [LSM] difference, 0.07%; 95% confidence interval: −0.07, 0.21). HbA1c was reduced by 0.56% and 0.63% with URLi and lispro, respectively, with no significant treatment difference (P = 0.321). URLi provided superior PPG excursion control versus lispro at 1 h (LSM difference: −14.6 mg/dL, P <0.001) and 2 h (LSM difference: −21.8 mg/dL, P <0.001) as well as other time points (30‒240 min) during the MMTT. Incremental area under the glucose curve during the MMTT was also significantly lower with URLi versus lispro. The safety profiles were generally similar between treatment groups.
In conclusion, URLi in a basal-bolus regimen demonstrated superior control of PPG and non-inferior HbA1c reduction compared with insulin lispro, with a comparable safety profile, in adult patients with T2D.
Source: Read Full Article