Study provides clues on curbing the aggressive nature of coronavirus
Recent study by Estonian researchers at the University of Tartu explains how coronavirus is activated before attacking cells and what could help to impede that. The study, published in Scientific Reports, takes us a step closer to understanding why the spread of SARS-CoV-2 has been so rapid and aggressive. The studied virus activation mechanism is also one potential target for developing drugs for the treatment of COVID-19.
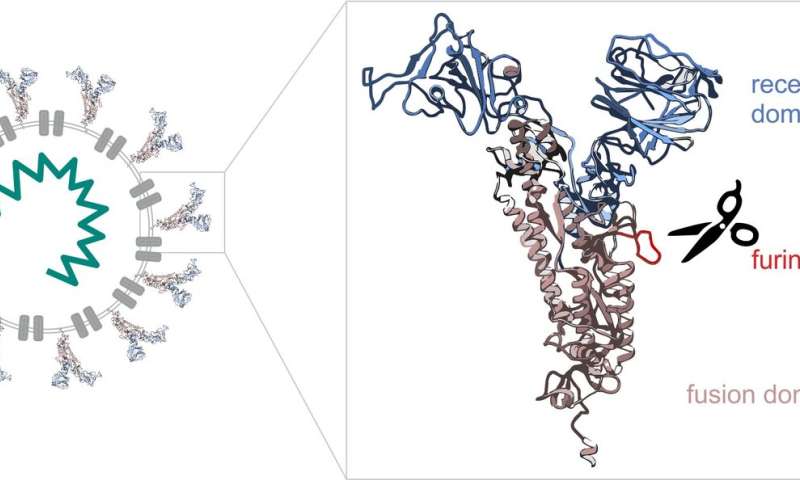
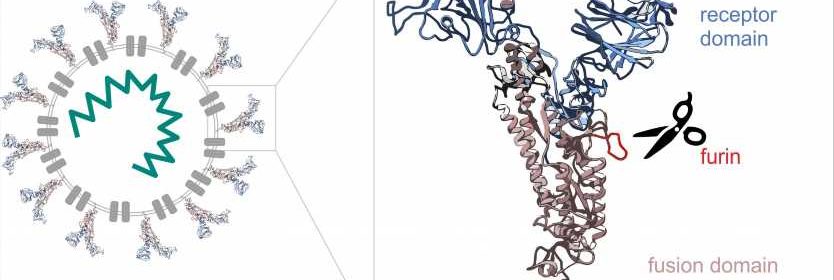
Molecular biologists Mihkel Örd and Ilona Faustova from the University of Tartu together with Professor Mart Loog compared the cell entry mechanisms of SARS-CoV-2 causing the current corona pandemic with those of other coronaviruses (SARS-CoV-1 and MERS-CoV causing the Middle East respiratory syndrome, with outbreaks in 2003 and 2012, respectively). The study focused on the spike protein that forms the spikes we know well from the images of the virus. The SARS-CoV-2 spike protein consists of two parts (see the Figure 1). The tip of the spike works like a detector, looking for a suitable cell to attach to. In cells, the exchange of information is mediated by various proteins that send and receive chemical signals. Viruses take advantage of these proteins to enter cells.
One protein the SARS-CoV-2 spike protein addresses is the enzyme furin that is present in many cells of the human body. Furin’s task is to cleave proteins, acting like biological scissors. Different enzymes affect different proteins. Among other proteins, furin also cleaves the SARS-CoV-2 spike protein, removing its detector-like tip. After that, the remaining part of the spike protein starts work by fusing with the cell membrane and allowing the virus to enter the cell.
“Many viruses use a similar logic. They have a receptor domain and a binding domain, but use different enzymes of the host. To some extent, this also affects the way the virus functions in the body. Different enzymes are expressed in different tissues. Furin is expressed throughout the human body, so the range of cells the virus can enter is enormous. We can suppose that this is why the virus is particularly aggressive,” explained Mihkel Örd, a doctoral student in biomedical technology at the University of Tartu (UT).
From cell division to taming the virus
In their everyday work, Mihkel Örd and UT Senior Research Fellow in Molecular Biology Ilona Faustova study what affects the division of cells and how. They pay special attention to phosphorylation—a chemical reaction in which a phosphoryl group is attached to a protein. As a result, the initial task or activity of the proteins may change, which allows controlling the intracellular processes in certain conditions.
The way enzymes process proteins depends on the patterns or short linear motifs of the amino acids in the protein. Proteins are composed of 20 different amino acids and the short linear motifs of amino acids may encode important biological information. When analyzing the sequence of the spike protein of the novel coronavirus, the researchers from Tartu soon noticed that in that critical site where the spike protein is supposed to be cut into two, there can be three motifs with a different task. When the spread of coronavirus turned into a pandemic, Örd and Faustova decided to study whether these predicted motifs function in real life and how they can affect the life cycle of the virus.
“We ordered DNA sequences of coronavirus, synthesized and separated the proteins from the cells, created mutations to detect furin-sensitive sites and to understand how phosphorylation affects them. There are more than one thousand amino acids in the spike protein, but for this process, only one tiny part is important. We devoted our attention to that site and started to study it in more detail,” Örd explained.
The analysis revealed that in the case of one motif, the furin cleavage site was very specifically encoded in the novel coronavirus. “What was surprising was how efficiently furin cleaved the SARS-CoV-2 spike protein motif. In comparison, we analyzed the motifs in viruses causing MERS and SARS that are only slightly different: furin failed to cleave them. Another interesting finding was that the cleavage site was surrounded by two other motifs the phosphorylation of which completely inhibited the furin cleavage of the spike protein. These findings provide new information about which processes may affect the life cycle of the virus and why the novel virus has been more successful than the previous two,” explained Faustova.
“In our research, we have always been guided by the idea that the better we understand different molecular mechanisms, the more possibilities we have to intentionally affect or control them. Since the studied motifs are repeated in different proteins, their description allows making predictions about the tasks and regulation of many proteins. The first published findings already confirm that furin inhibitors curb the replication of SARS-CoV-2 and may have a potential pharmaceutical application,” said Faustova.
Corona crisis put the research system to a test
Faustova adds that the corona pandemic has revealed the collaboration capacity of the global research community. “We have studied the encoded enzyme signals of different motifs for years and this allowed us to contribute to understanding the mechanisms of coronavirus based on our expertise. Hundreds of labs across the world have done the same. In the crisis, other projects were halted to jointly focus on the new virus. Over the past nine months, life scientists of the world have made enormous progress. Never before have we managed to join all of our forces and conduct a detailed study of a single critical process. High-quality publications in the field are published every day. This gives us hope that the corona vaccine and drugs may reach us pretty soon.”
Source: Read Full Article