Clinical and epidemiologic features of SARS-CoV-2 in companion animals

In a recent study posted to the Research Square* preprint server, researchers explored the clinical and epidemiologic characteristics of coronavirus disease 2019 (COVID-19) in dogs and cats.
Numerous beneficial effects of pet ownership, including benefits to mental health, have been scientifically established. Companion animals account for 60% of all animals reported to World Organization for Animal Health (WOAH) globally between 29 February 2020 and 31 December 2021, making them the second-most often reported animal group to be infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) after farmed mink. At the national and international levels, however, there are limited surveillance efforts to identify zoonotic disease transmission of pathogens, such as SARS-CoV-2, in companion animals.

About the study
In the present study, the researchers from theU.S. Centers for Disease Control and Prevention (CDC), the U.S. Department of Agriculture (USDA), and Texas A&M University described the clinical and epidemiologic characteristics of SARS-CoV-2 in companion animals in the U.S. that have been discovered through both passive and active surveillance.
Animal SARS-CoV-2 cases are detected in the U.S. by either passive or aggressive surveillance. For example, when pet owners bring their companion animals to veterinary clinics or hospitals, case identification is often started via passive surveillance. Samples are then subsequently sent to various veterinary diagnostic laboratories for SARS-CoV-2 testing. In addition, animals with known SARS-CoV-2 exposure or clinical symptoms suggestive of SARS-CoV-2 infection are actively sought by health officials or researchers through active surveillance. These include joint One Health studies of SARS-CoV2 transmission in households, zoos, animal shelters, animal rescues, animal rehabilitation facilities, and veterinary clinics involving both people and animals.
As per the U.S. case definition, an animal is ascertained to be SARS-CoV-2 positive at the United States Department of Agriculture National Veterinary Services Laboratories (USDA-NVSL) if either of the following conditions is true: (1) the SARS-CoV-2 sequence was generated directly from a suspect or presumptive positive animal specimens or indirectly from a viral isolate obtained from that animal, or (2) serological sample from a suspect or presumptive positive animal showed the presence of SARS-CoV-2 neutralizing antibody.
Any dog and cat samples that fulfilled the following requirements were required to be submitted to the USDA-NVSL: (1) strong sequencing candidates having SARS-CoV-2 real-time reverse transcription–polymerase chain reaction (RT-PCR) cycle threshold (Ct) values less than 30; (2) linked to unusual morbidity and mortality events; or (3) suspected or known to be infected with SARS-CoV-2 variants including Alpha, Delta, and Omicron variants.
Based on the body systems that were affected, the researchers divided the clinical indicators into three categories: respiratory signs including cough, shortness of breath, difficulty breathing, sneezing, nasal discharge, and ocular discharge, gastrointestinal symptoms including vomiting and diarrhea, and non-specific symptoms such as lethargy, inappetence, fever). In addition, ct values and virus neutralization (V.N.) titers were measured following exposure to a COVID-19-infected person to understand the timeline of infection and immune response of companion animals infected with SARS-CoV-2.
Results from the SARS-CoV-2 RT-PCR and SARS-CoV-2 VN titers were compared to those observed at the alleged date of exposure. The geometric mean titer and lowest Ct values among oral and nasal swabs were calculated every two days. For animals confirmed to be COVID-19-infected, average Ct values and log-transformed geometric mean viral neutralizing antibody titers were determined.
Results
Approximately 345 animals observed in 33 states in the U.S. tested positive for SARS-CoV-2 between March 2020 and December 2021. Almost 59% of them, including 109 cats and 95 dogs, were companion animals, while 94% of the companion animal cases discovered through passive surveillance involved initial contact with a COVID-19-positive person.
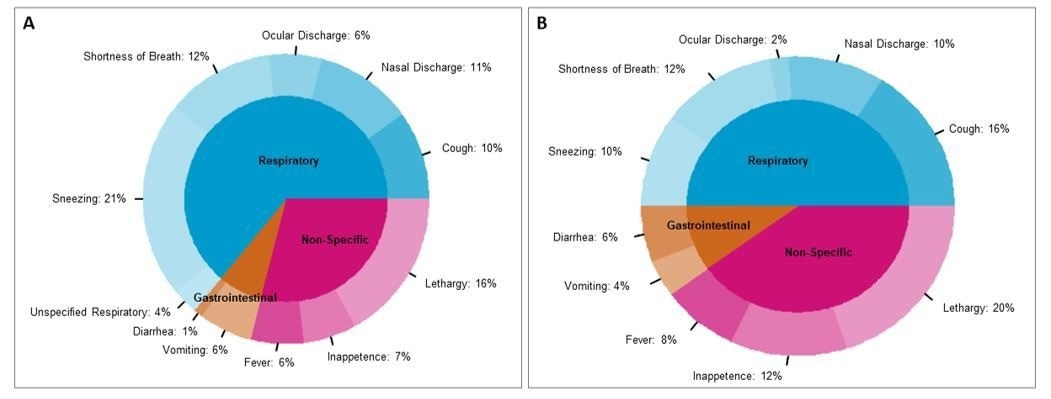
Clinical signs reported in cats (n=55; A) and dogs (n = 42; B). Of 97 animals with clinical signs, the proportion of each clinical sign being displayed are shown within each species. Since a given animal may display multiple clinical signs, percentages are calculated by number of signs displayed, not by individual animals.
The study results showed that 52% of companion animals had no documented clinical indications at the time of sampling, while 48% had clinical signs consistent with SARS-CoV-2 infection. This differed significantly depending on the type of surveillance used; although 72% of animals discovered by passive surveillance showed clinical symptoms, only 27% of those discovered through active surveillance reported signs.
Animals exhibiting clinical signs most typically were reported as having respiratory symptoms as compared to non-specific and gastrointestinal symptoms. Among the animals with clinical symptoms, 21% and 16% of the cats experienced sneezing and lethargy, while 16% and 20% of the dogs had cough and lethargy, respectively.
Among the 204 confirmed COVID-19-positive companion animals, 45% tested positive by V.N. alone, 33% by only RT-PCR, and 23% by both V.N. and RT-PCR. For all companion animals that were proven positive using RT-PCR data, the average Ct value was 28.6. All confirmed COVID-19-positive companion animals had median V.N. titers of 64, ranging from 8 to 512. Among these, titers from dogs ranged between 8 to 128 with a median titer of 32, while those from cats ranged between 32 to 512 with a median titer of 128.
Conclusion
The study findings showed that companion animals like dogs and cats are infected with SARS-CoV-2 after exposure to a COVID-19-infected individual who is often their owner. Therefore, case studies and surveillance involving both humans and animals are essential to comprehend the transmission and the viral development of zoonotic illnesses like SARS-CoV-2.
*Important notice
Research Square publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
- Amanda Y. Liew, Ann Carpenter, Taylor A. Moore et al. Clinical and epidemiologic features of SARS-CoV-2 in dogs and cats compiled through national surveillance in the United States, 16 September 2022, PREPRINT (Version 2) available at Research Square, DOI: https://doi.org/10.21203/rs.3.rs-1987536/v2, https://www.researchsquare.com/article/rs-1987536/v2
Posted in: Medical Science News | Medical Research News | Disease/Infection News
Tags: Agriculture, Antibody, Breathing, Coronavirus, Coronavirus Disease COVID-19, Cough, covid-19, CT, Diagnostic, Diarrhea, Fever, Immune Response, Lethargy, Mental Health, Mortality, Omicron, Polymerase, Polymerase Chain Reaction, Research, Respiratory, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Sneezing, Syndrome, Transcription, Veterinary, Virus, Vomiting

Written by
Bhavana Kunkalikar
Bhavana Kunkalikar is a medical writer based in Goa, India. Her academic background is in Pharmaceutical sciences and she holds a Bachelor's degree in Pharmacy. Her educational background allowed her to foster an interest in anatomical and physiological sciences. Her college project work based on ‘The manifestations and causes of sickle cell anemia’ formed the stepping stone to a life-long fascination with human pathophysiology.
Source: Read Full Article