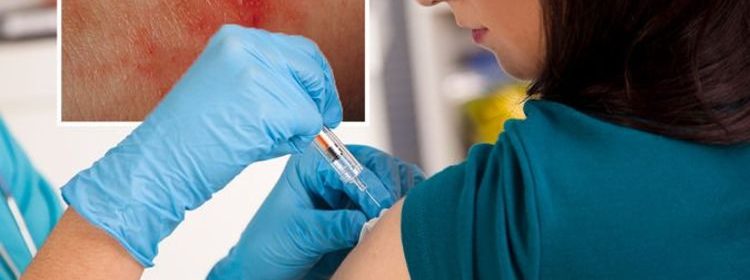
AstraZeneca vaccine: ‘Pin-prick rash’ and six other side effects that require medical help
Matt Hancock grilled by Nick Ferrari on ‘drop’ in administered vaccines
When you subscribe we will use the information you provide to send you these newsletters. Sometimes they’ll include recommendations for other related newsletters or services we offer. Our Privacy Notice explains more about how we use your data, and your rights. You can unsubscribe at any time.
The AstraZeneca vaccine rollout in Europe has been beset by problems, chief among them is the perceived threat of blood clots following vaccination. Ongoing investigations are seeking to establish the scale of the threat, although the UK’s regulatory body has so far concluded that blood clots are an “extremely rare” occurrence.
While the Medicines and Healthcare products Regulatory Agency (MHRA) continues to investigate these cases, as a precautionary measure anyone that develops symptoms after vaccination is advised to seek prompt medical advice.
Public Health England (PHE) has listed a number of symptoms that should prompt medical attention.
According to PHE, unexplained pin-prick rash or bruising beyond the injection site should be checked out by a health professional.
Other serious side effects include:
- Shortness of breath
- Chest or persistent abdominal pain
- Leg swelling
- Blurred vision
- Confusion or seizures.
“Furthermore anybody with new onset of severe or persistent headache that does not respond to simple painkillers starting four days or more after vaccination should speak to their doctor,” adds PHE.
Blood clot – UK latest
The MHRA has undertaken a thorough review into UK reports of an extremely rare and unlikely to occur specific type of blood clot in the brain, known as cerebral venous sinus thrombosis (CVST) occurring together with low levels of platelets (thrombocytopenia) following vaccination with the COVID-19 Vaccine AstraZeneca.
It is also considering other blood clotting cases (thromboembolic events) alongside low platelet levels.
On the basis of this ongoing scientific review, it has concluded that the evidence of a link with COVID-19 Vaccine AstraZeneca is stronger but more work is still needed.
DON’T MISS
Pfizer vaccine: South African Covid variant can ‘break through’ [INSIGHT]
Statins side effects: Best and worst foods [ADVICE]
AstraZeneca vaccine may be linked to capillary leak syndrome [INSIGHT]
“As a precaution, administration of COVID-19 Vaccine AstraZeneca in people of any age who are at higher risk of blood clots because of their medical condition should be considered only if benefits from the protection from COVID-19 infection outweighs potential risks,” advises PHE.
According to the health body, anyone who experienced cerebral or other major blood clots occurring with low levels of platelets after their first vaccine dose of COVID-19 Vaccine AstraZeneca should not have their second dose.
“Anyone who did not have these side effects should come forward for their second dose when invited,” it adds.
The MHRA recently confirmed that the evidence to date does not suggest that the COVID-19 Vaccine AstraZeneca causes venous thromboembolism without a low platelet count.
Everyone should continue to get their vaccination when asked to do so unless specifically advised otherwise, advises the UK drug body.
How many people in the UK have now been vaccinated?
According to the latest figures, more than 32 million people in the UK have had a first dose of a coronavirus vaccine.
According to public health figures, a total of 32,121,353 have received their first dose of a vaccine.
The figures show that a total of 7,466,540 people have received their second dose of a vaccine.
The NHS is currently offering the COVID-19 vaccine to people most at risk from coronavirus.
In England, the vaccine is being offered in some hospitals and pharmacies, at local centres run by GPs and at larger vaccination centres.
More centres are opening all the time.
The order in which people will be offered the vaccine is based on advice from the Joint Committee on Vaccination and Immunisation (JCVI).
Source: Read Full Article